The Mako Difference
Mako Total Knee can help lead to better outcomes, like less pain1 and shorter recovery times1, compared to manual surgery.
In clinical studies, compared to manual knee replacement surgery, Mako Total Knee:
-
Enabled surgeons to execute their surgical plans more.2,3
-
Protected soft tissue and ligaments from damage.4,6
-
Resulted in lower pain scores among patients surveyed 6 months after surgery than those who received conventional joint replacement.5
-
Resulted in better patient satisfaction scores among patients surveyed 6 months after surgery compared to those who received a conventional joint replacement.5
Learn more about Mako Robotic-Arm assisted surgery.
Discover Mako SmartRobotics™ surgeons near you to see if a Mako Knee procedure is an option for you.
The journey of Mako Total Knee Replacement (TKA) surgery
Step 1 - Scan
Before surgery, a CT scan of your knee is taken and used to develop a 3D virtual model of your unique joint.

Step 2 - Plan
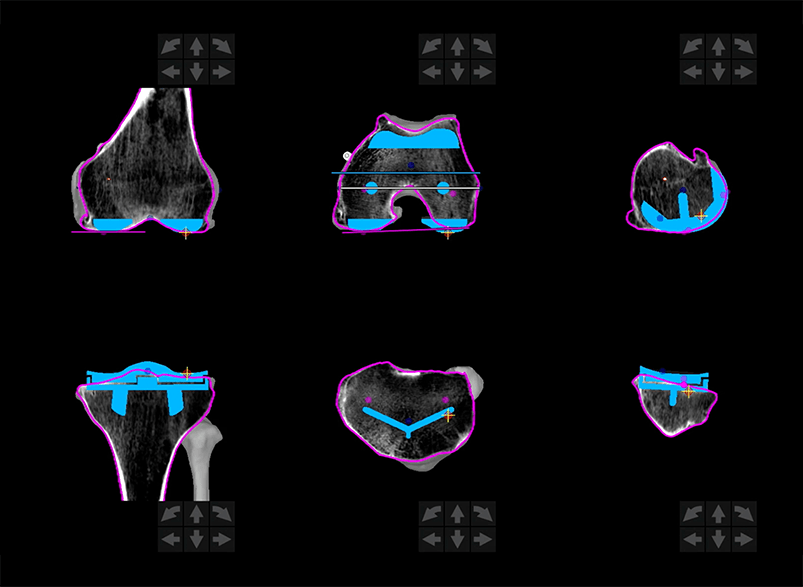
The second step is patient-specific surgical planning.
Your doctor uses the 3D CT-based model to evaluate your bone structure, disease severity, joint alignment and even the surrounding bone and tissue so that they can determine the desired size, placement and alignment of your implant.
Step 3 - Mako Can
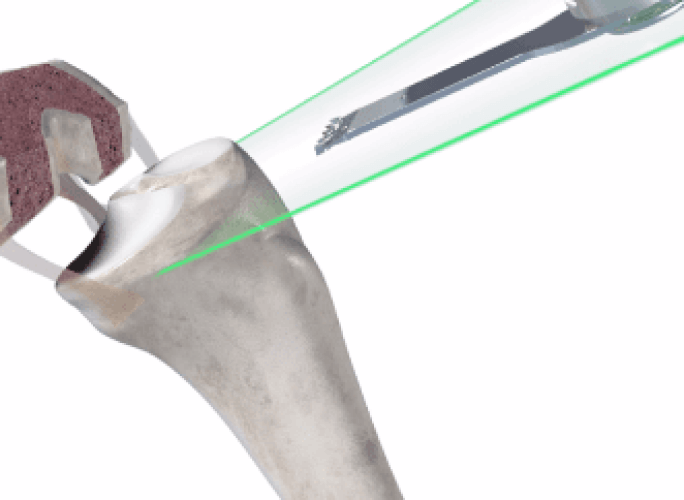
In the operating room, your surgeon guides Mako’s robotic arm to remove arthritic bone and cartilage from the knee.
Mako’s Accustop™ technology creates a virtual boundary that provides tactile resistance to help the surgeon stay within the boundaries defined in your surgical plan.
Throughout the procedure, Mako provides real-time data to your surgeon, allowing them to assess the movement and tension of your joint and adjust your surgical plan if desired.
Is Mako right for me?
Mako Total Knee Arthroplasty is for people who:
-
Experience severe knee pain or stiffness resulting from: noninflammatory degenerative joint disease (including osteoarthritis, traumatic arthritis, or avascular necrosis), rheumatoid arthritis or post-traumatic arthritis.
-
Haven't experienced adequate relief with conservative treatment options, like bracing, medication or joint fluid supplements.