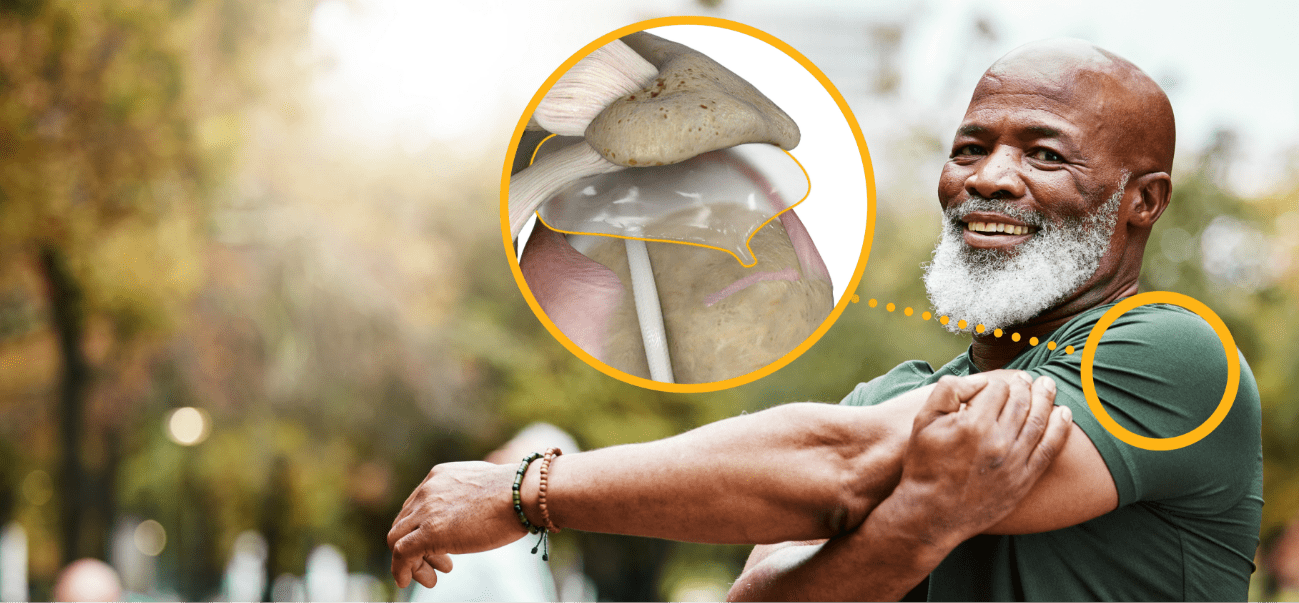
The InSpace balloon

What is the InSpace balloon?
The InSpace balloon is a treatment option for people 65 years old or older who have a massive irreparable rotator cuff tear and may benefit from the device based on evaluation by a surgeon.
To date, over 41,000 InSpace balloon procedures have been performed worldwide. Surgery with the InSpace balloon can offer patients2:
- Pain relief.
- Increased range of motion.
- Early return to shoulder function.
How does the InSpace balloon work?
The InSpace balloon is placed under your shoulder bone during minimally invasive arthroscopic surgery. This helps decrease rubbing between your arm bone and your shoulder bone to help relieve pain and restore function.
After surgery, the InSpace balloon breaks down in your body over time and has been shown to completely break down after about one year.
Watch the video below to see what happens during an InSpace balloon surgery.
The InSpace balloon offers a simple and minimally invasive treatment option with long-term safety and results similar to a partial repair surgery which is a commonly performed procedure.1
Please note that while many patients may experience benefits from treatment with the InSpace balloon, good results after InSpace balloon surgery are not guaranteed. Please refer to “Important Information” below for further details on risks.
Discuss treatment options with your surgeon to determine if the InSpace balloon is right for you.
If you’re interested in learning more about the InSpace balloon, visit the InSpace resources page
Find a surgeon who offers the InSpace implant
FIND A SURGEONReferences:
- Data on file from Stryker.
- Verma N, Srikumaran U, Roden CM, Rogusky EJ, Lapner P, Neill H, Abboud JA. (2022). InSpace implant compared with partial repair for treatment of full-thickness massive rotator cuff tears. J Bone Joint Surg Am. Advance online publication. doi. 10.2106/JBJS.21.00667.
IMPORTANT INFORMATION ABOUT INSPACE
The InSpace Implant is indicated for the treatment of patients with massive, irreparable full-thickness torn rotator cuff tendons due to trauma or degradation with mild to moderate gleno-humeral osteoarthritis in patients greater than or equal to 65 years of age whose clinical conditions would benefit from treatment with a shorter surgical time compared to partial rotator cuff repair.
The InSpace Implant is not appropriate for patients with certain types of infections, known or suspected allegories to the implant and/or instrument materials, or children.
The following information provided is based on data collected in the US Pivotal Clinical Study. There is evidence of increased risk of total adverse events of the affected shoulder compared to a partial rotator cuff repair, which were reported to be mostly mild to moderate in severity. Please note that complications reported by InSpace and Partial Repair patients were reviewed by a group of experts in shoulder surgery, and it was determined that none were related to the treatment devices (i.e., the InSpace Implant or devices used for Partial Repair). There is evidence of increased adverse events associated with a serious risk of the index shoulder which includes both medical and surgical treatments. The InSpace Implant is not fixed in place and may change position in the shoulder space. Movement out of the subacromial space and/or deflation of the InSpace Implant was observed in magnetic resonance imaging (MRI) 6 weeks post-operatively in up to half of patients. In these cases, it should be noted that no re-operations were required to remove the InSpace Implant.
The risks and benefits of the InSpace Implant in patients with the following conditions should be carefully considered: severe arthritis in the shoulder or full thickness cartilage loss, deltoid muscle defect or deltoid muscle paralysis, evidence that the shoulder is very unstable, missing or non-intact ligaments in the shoulder, known loss of sensation, major medical conditions such as blood clotting disorders, conditions that suppress the immune system or any other conditions that may compromise healing, safety and effectiveness of InSpace Implant use in patients below age sixty-five (65) has not been established, and safety and effectiveness of InSpace Implant use in pregnant or nursing mothers has not been evaluated.
If a reaction occurs, it will require follow-up with your surgeon. Some examples are as follows: irritation at the site where a cut was made during surgery, local infection, inflammation, and tissue damage where the InSpace Implant was implanted, or worsening of an existing infection. Additionally, movement of the implant out of its intended location may lead to worsening of your shoulder pain, discomfort, and/or stiffness which may require a re-operation, narcotic medications, non-narcotic pain medications, or post-operative shoulder injections.
As with any surgery, your surgical procedure may cause complications or adverse events. As with any shoulder surgery involving anesthesia there are potential risks and complications, including dizziness, fainting, difficulty breathing, wound infection, wound drainage, swelling, local pain, bleeding, bruising, surgical wound opening, nerve pain or injury which could result in partial or complete loss of movement in your affected arm, tendon injury, loss of sensation in your skin and muscle around your surgical site, inflammation of the tissues that surround your rotator cuff joint, a frozen rotator cuff (inability to freely move the shoulder joint) and delayed wound healing. There is a risk that a deep vein clot will form (thrombosis) and travel to the lung (pulmonary embolism), which could lead to stroke, heart damage or death. Any of the aforementioned complications/risks could require treatment such as medicine or additional surgery that may involve replacing or removing your InSpace Implant dependent on timeframe as the implant is biodegradable.
The information presented is for educational purposes only. Speak to your doctor to decide if you are interested in learning more about the InSpace Implant. Individual results vary and not all patients will receive the same postoperative activity level. It is important to follow your surgeon’s advice regarding after surgery care (for example, physical therapy) as well as which immediate activities you may or may not perform after receiving the InSpace Implant. Not following this advice may cause complications with the InSpace Implant. Some complications may require you to have additional surgery. Ask your doctor if the InSpace Implant is right for you.
Stryker Corporation or its other divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: InSpace, Stryker, Together with our customers, we are driven to make healthcare better. All other trademarks are trademarks of their respective owners or holders.
1000904124 Rev B